Global Lessons for Controlling Healthcare Costs
Health insurers are reaching beyond national borders for more effective medical management strategies.
Health insurers — the private companies and government healthcare payors that foot the majority of the US$7 trillion global healthcare bill — have a rather insular heritage. Because the structures and mandates of national healthcare systems vary widely, payors in one system have often presumed that they had little in common with payors elsewhere. But that is increasingly changing.
A flurry of activity is evident as more and more payors in both developed and developing nations reach beyond their borders. They are seeking proven treatment guidelines and attempting to utilize reference standards created in other healthcare systems. They are also investing internationally to enhance their revenue streams and gain access to broader practices and knowledge.
The main impetus for this outreach is the recognition that healthcare payors all have an urgent need to find more effective medical management strategies — i.e., those capable of improving patient access to care and care outcomes, while also bringing healthcare costs under control. Of course, some payors are part of systems that are performing better than others in these ways, but all systems face challenges — and none of them have fully figured out the formula for providing high-quality, universal care at a cost that is sustainable over the long haul.
Too Big a Burden
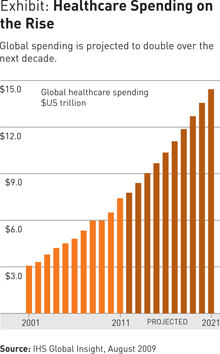
The quest for this formula is especially intense among payors because of the growing financial burden they are being asked to shoulder as spending in the healthcare sector continues its rise. (See Exhibit.) This burden is being driven by two trends: the ever-increasing cost of medical treatment and rising demand for care.
Cost. The cost of care, which is rising faster than the consumer price index in many national healthcare systems, is a complex function of multiple factors, such as medical innovation, rising provider costs, increasing prevalence of expensive chronic conditions, and tightened capacity, to name a few. From the pharmaceutical perspective, a number of blockbuster drugs are becoming available in low-cost generic versions, but many of the highly effective new treatments being produced in the biotechnology sector are coming to market at 10 to 20 times the cost of conventional pharmaceuticals. For example, the use of biomarkers enables drugmakers to customize their offerings for more focused patient populations. But this also means that pharmaceutical companies’ investments must be recouped over a smaller base, and prices thus need to rise. At the same time, new and technologically sophisticated diagnostics and equipment are raising the cost of medical tests and procedures.
Not only do these medical advances carry a higher sticker price, but they also create additional costs down the road. Their very effectiveness can lead to the so-called chronification of diseases that were once killers. In the process of saving lives, these powerful medicines and tools create prolonged courses of treatment that add greatly to the overall lifetime cost of care. For example, Genentech’s cancer drug, Avastin, can cost $4,000 to $8,000 monthly, and it can extend patients’ lives by as much as two years.
Demand. The other great source of instability in healthcare systems is the explosion in demand. In many developed nations, populations are aging, and older people, who consume healthcare at much higher rates than any other age demographic, are living longer. For example, in the U.S., in 2006, the Center for Retirement Research of Boston College reported that annual medical costs for people age 55 to 64 are nearly four times as high as for people age 35 to 44.
Developing nations, especially those with emerging economies, face a different challenge. Their populations are younger and expanding, and per capita income is rising — and so is the incidence of the chronic diseases of the West, such as diabetes. This translates into greater demand for healthcare. In China, for example, healthcare expenditures are expected to grow by 18 percent annually over the next several years.
Rising costs and rising demand have been exacerbated by the inability of governments to fund healthcare systems. The global economic turmoil that began in 2008 continues to reverberate in the form of individual national crises and a slow, halting recovery.
The magnitude of the challenges that these conditions are creating varies among healthcare systems. Former Canadian Prime Minister Brian Mulroney recently pointed out that his country’s government-funded universal healthcare system is not financially sustainable. “We face a crisis if efforts are not made to avert disaster. Health-care costs are exploding, increasing by 5 per cent to 7 per cent annually. Yet, GDP and tax revenue — which finance health care — are growing much more slowly,” he wrote in an editorial in the Globe and Mail. “At this rate, health care will make up 80 per cent of total program spending in Ontario by 2030, leaving all other programs to be funded from the remaining 20 per cent.… This same trend can be seen across all the provinces and territories.”
Canada isn’t alone. In the U.S., Medicare went “cash-flow negative” in 2008. And even though the reforms in the 2010 Affordable Care Act improved the outlook, the boards of trustees of the Social Security and Medicare trust funds still report that the Hospital Insurance Trust Fund will be insolvent by 2029.
The bottom line: For many healthcare systems, and the payors within them, the challenges of cost and demand have created a looming crisis and have given rise to the specter of systemic collapses.
Searching for Sustainability
All healthcare systems are facing challenges in terms of sustainability. But the value for payors in taking a global approach in their quest for more effective medical management strategies lies as much in the diversity of healthcare systems as in their commonalities. Simply put, incremental approaches in siloed national markets are unlikely to be enough to rebalance the cost equation.
The structures and payment schemes within systems vary widely, as do the development levels of their care-delivery infrastructure and care provider bases, and the needs of the people they serve. This has led to a broad range of solutions to the issues of cost and demand.
More and more payors are reaching outside their home systems to explore and capture the value these solutions offer. Their efforts take three common forms.
1. Treatment guidelines. Treatment guidelines are used by doctors and hospitals to standardize care, improving outcomes as well as reducing ineffective treatment, medical errors, and waste. These actions translate directly into lower costs for payors, which are increasingly encouraging (and, in some cases, measuring) guideline development and adoption.
So many government payors were interested in creating and implementing clinical guidelines that in 2008, the U.K.’s National Institute for Health and Clinical Excellence, which develops guidelines for the National Health Service, formed NICE International. NICE International is a nonprofit organization whose goal is to help develop healthcare systems globally. Its work is funded by government agencies and international organizations, such as the World Bank and the World Health Organization. And although NICE was developed for application in the U.K., its impact is broadening as other systems adopt NICE recommendations and engage its capabilities directly.
In 2009, for example, the ministry of health in the Indian state of Kerala, which funds public healthcare, tapped NICE International for its expertise in clinical guideline development. Kerala has a healthy population and a well-educated base of doctors, but the ministry of health is seeking to stem the decline in some of the state’s health indicators by learning how to more effectively develop and implement guidelines, especially for rural clinics and for diseases, such as leptospirosis, that carry a high burden of care, but are relatively simple to treat. NICE International has also worked with the ministries of health in China, South Africa, Turkey, and many other nations.
This trend is not restricted to developing nations. Payors and pharma companies in the U.S. are showing significant interest in designing comparative effectiveness into the clinical trials for new therapies. Comparative effectiveness research, which is commonly used in the European Union and is growing in use in the U.S., seeks to determine how a given therapy ranks against alternative therapies and, thus, determine whether it represents a true improvement in cost-effective care. Comparative effectiveness is one key criterion to determine drug reimbursement prices under the most recent German healthcare reform, AMNOG. In the past, the German system allowed “free pricing” (although in truth many measures to contain drug costs and prices had been used before). Today, however, the principle of value-based pricing is the key paradigm. The new approach takes into account the medical outcome of the drug’s studies, the time of the drug’s introduction (assuming comparable efficacy of the followers, only the first drug in its class that makes it to market will get the full price for innovation), and comparative costs (costs compared with existing standard therapy costs per day per patient).
Government agencies from widely diverse health systems are increasingly looking across national borders for lessons on improving the access/care/cost equation, whether in the form of government actions or enhanced private-system approaches intended to supplement the public model.
2. Direct reference. Direct reference is an economical means of improving care and reducing costs by adopting proven medical standards and/or pricing models developed in other healthcare systems or by international organizations. As a result, the adopting payors can avoid the infrastructure and cost associated with establishing standards.
A good of example of direct reference can be found in Europe, where Germany and the United Kingdom use value-based approaches to setting pharmaceutical prices. In turn, a number of other European nations simply adopt those pricing structures based on a price basket that references the drug reimbursement prices in Germany, the U.K., or both. Turkey uses a reference pricing system, in which a basket of the drug’s pricing in France, Spain, Italy, Portugal, and Greece, as well as the country of production and country of export, are considered in setting price ceilings and floors.
This trend has significant implications for pharma companies. For example, as certain countries encounter serious economic stress, some are taking less nuanced approaches to across-the-board price cuts, which could be emulated more broadly: In December 2010, Greek hospitals began issuing non-interest-bearing bonds for payment to suppliers; additionally, Greece has both issued stiff mandatory reimbursement price cuts in 2010 and put growth limits on drug prices by cutting by 2.5 percent the prices for new drugs whose sales grew by more than 5 percent in one year.
The widespread adoption of ICD-10 codes by countries is another example of direct reference. ICD-10 (the 10th revision of the International Statistical Classification of Diseases and Related Health Problems) was developed under the auspices of the World Health Organization. It is a standardized system of codes for diseases and other health problems. These codes facilitate the statistical analysis of illness, which enables payors to better control costs and improve care. They also enable faster, more accurate provider reimbursement. ICD-10 is used in more than 100 countries in the reporting of health statistics; it is used by payors in at least 25 countries to determine resource allocation and reimbursement.
3. Direct investment. The third common form of global outreach is direct investment by private insurers. Insurers are seeking not only to leverage their existing capabilities and capture new revenue streams, but also to gain access to the practices and knowledge in these markets.
Like many private insurers, U.S.-based Aetna Inc. has used health plans for expatriates as a platform for international expansion. Aetna Global Benefits (AGB) provides health coverage for more than 325,000 expatriates, third-country nationals, and key local nationals in more than 100 countries. In 2008, AGB established an office in Shanghai, its first step into China’s growing medical insurance market.
Aetna is also leveraging its capabilities overseas. Its U.K. subsidiary, Aetna Health Services, provides care-management and demand-management services, as well as information services, such as data management, needs analysis, predictive modeling, and clinical decision support technologies. In January 2011, the subsidiary won an award from the British magazine Health Service Journal for its role in a chronic care program involving primary care doctors in the West Midlands that reduced the hospital admissions among participating patients by 45 percent.
UnitedHealth International (UHI), the global arm of U.S.-based UnitedHealth Group, offers Global Choice, a comprehensive health plan developed by British insurer BUPA International. BUPA is the largest player in the expatriate market, insuring more than 8 million expatriates in 190 countries.
UHI also partners with selected players in other countries to help them improve care and reduce costs by blending local needs with lessons learned from the U.S. healthcare industry. For example, since 2004, UHI has been working with the U.K.’s National Health Service to implement its Evercare program, a U.S. model that uses proactive care delivered by nurses to keep older people healthy and out of hospitals.
In and of themselves, treatment guidelines, direct reference, and direct investment by payors will not solve all the problems of failing or inefficient healthcare systems. But it is clear that the payors and the healthcare systems they operate within can benefit from a more global approach to solving their problems and securing their future. Just as consumer and industrial companies can achieve better results by taking a broad-based approach to generating new product ideas, healthcare payors have a far better chance of developing more effective medical management strategies if they cast a wider net. They won’t pull in winning ideas with every cast, but they will certainly have better catches than payors who fish in only one pond.![]()
Author profiles:
- Peter Behner is a partner with Booz & Company and the firm’s European health practice leader. Based in Berlin, he focuses on growth strategies, distribution channel management, and logistics optimization in the pharmaceutical industry.
- Rick Edmunds is a senior partner and global health practice leader for Booz & Company in Washington, D.C. His focus is corporate and commercial strategies for life science companies and U.S. payors.
- Elizabeth Powers is a principal in Booz & Company’s healthcare practice in New York, specializing in pharmaceutical commercial strategy.
- Also contributing to this article was Booz & Company senior associate Nelia McGreevy.